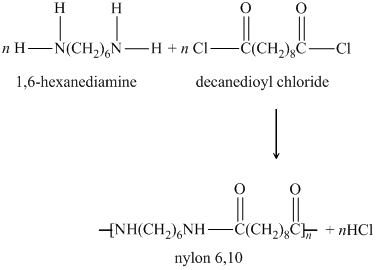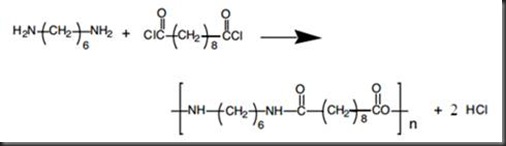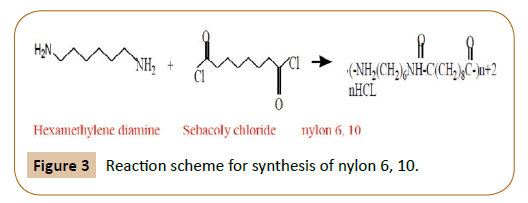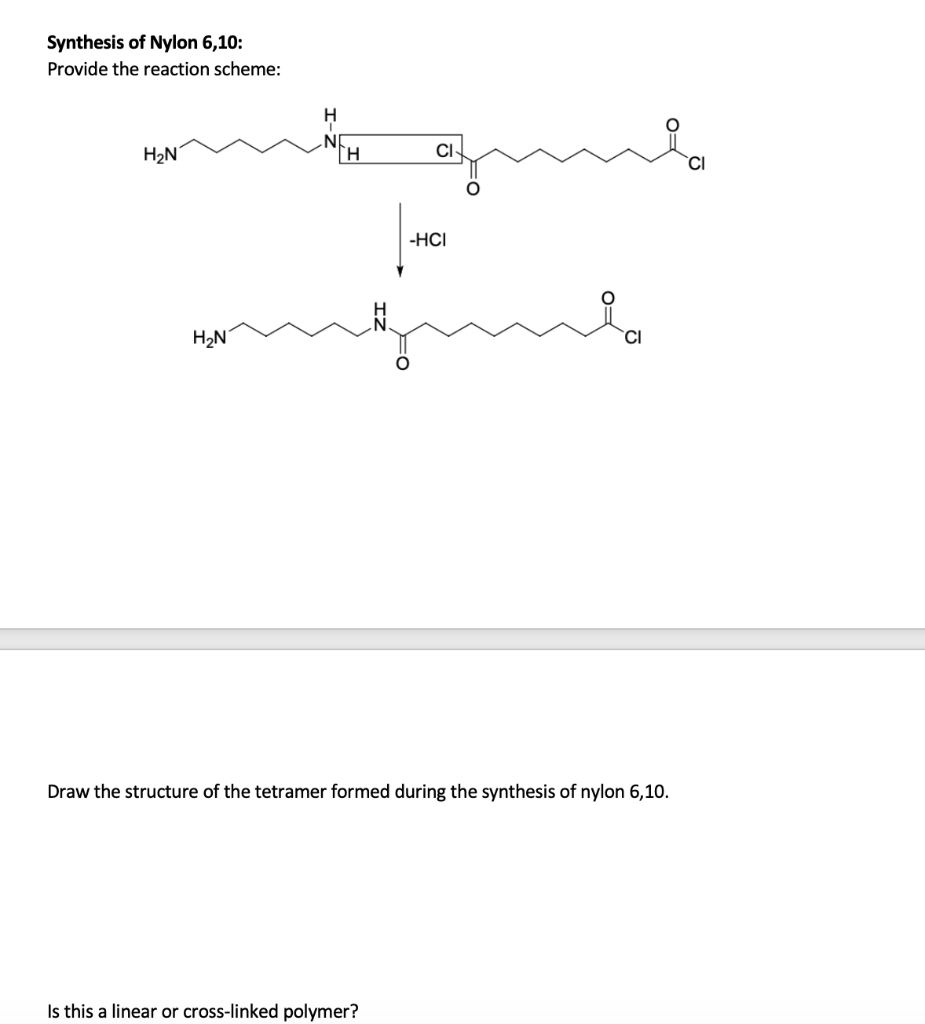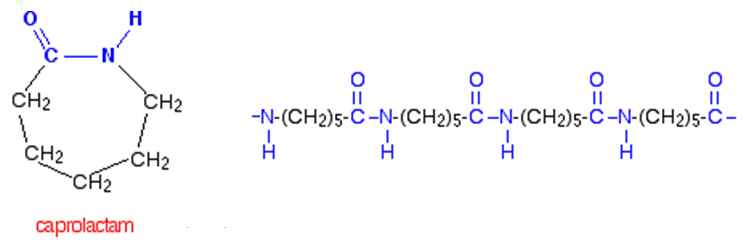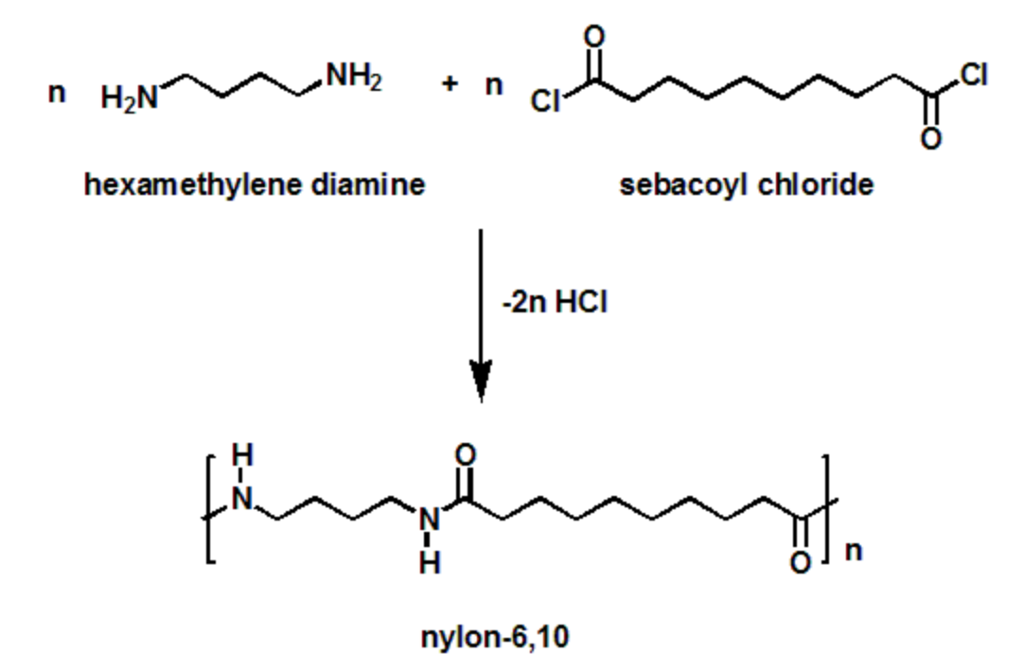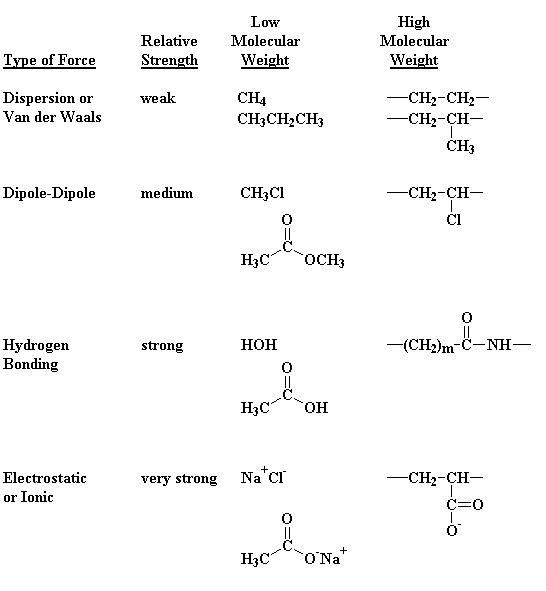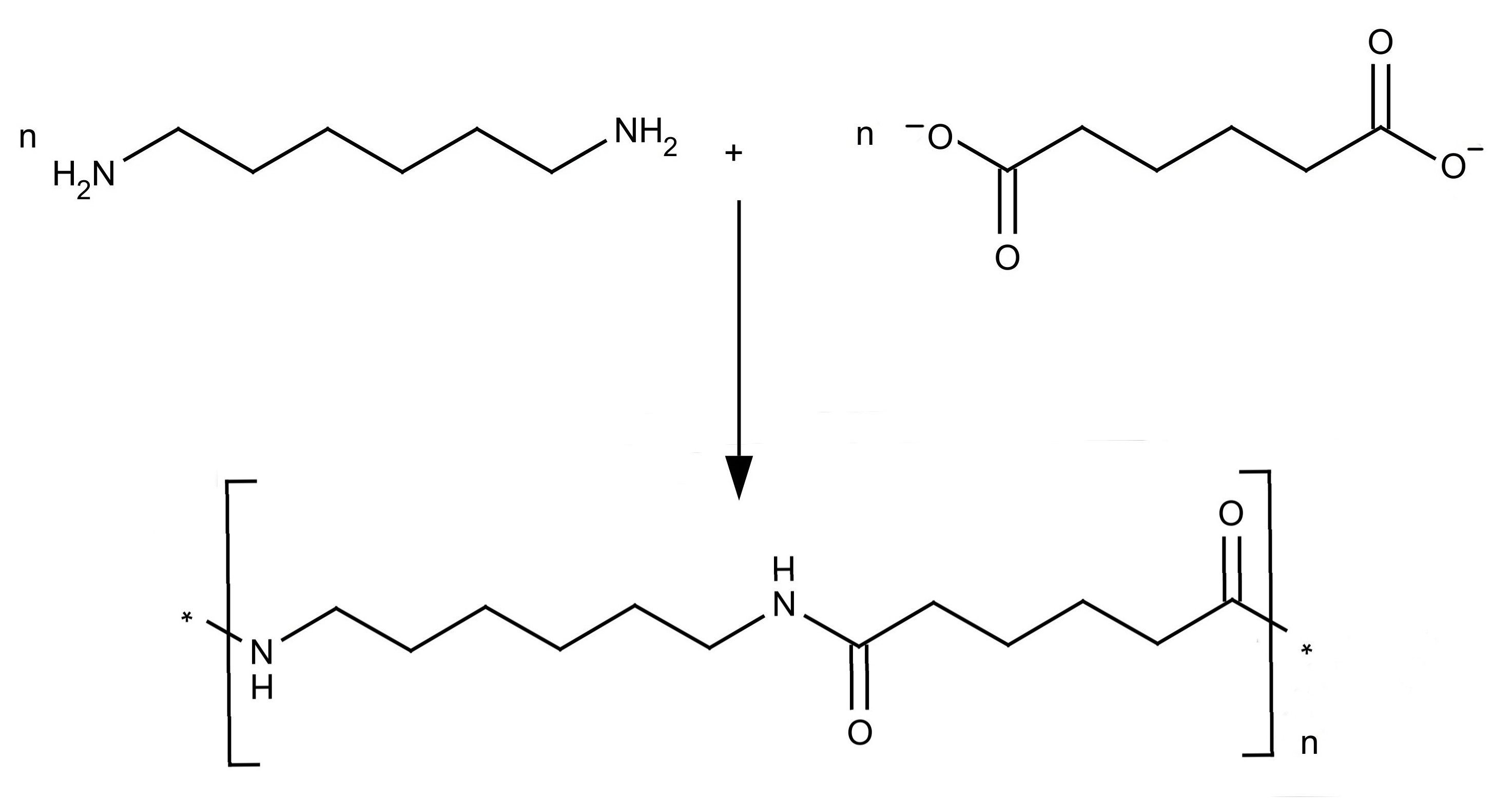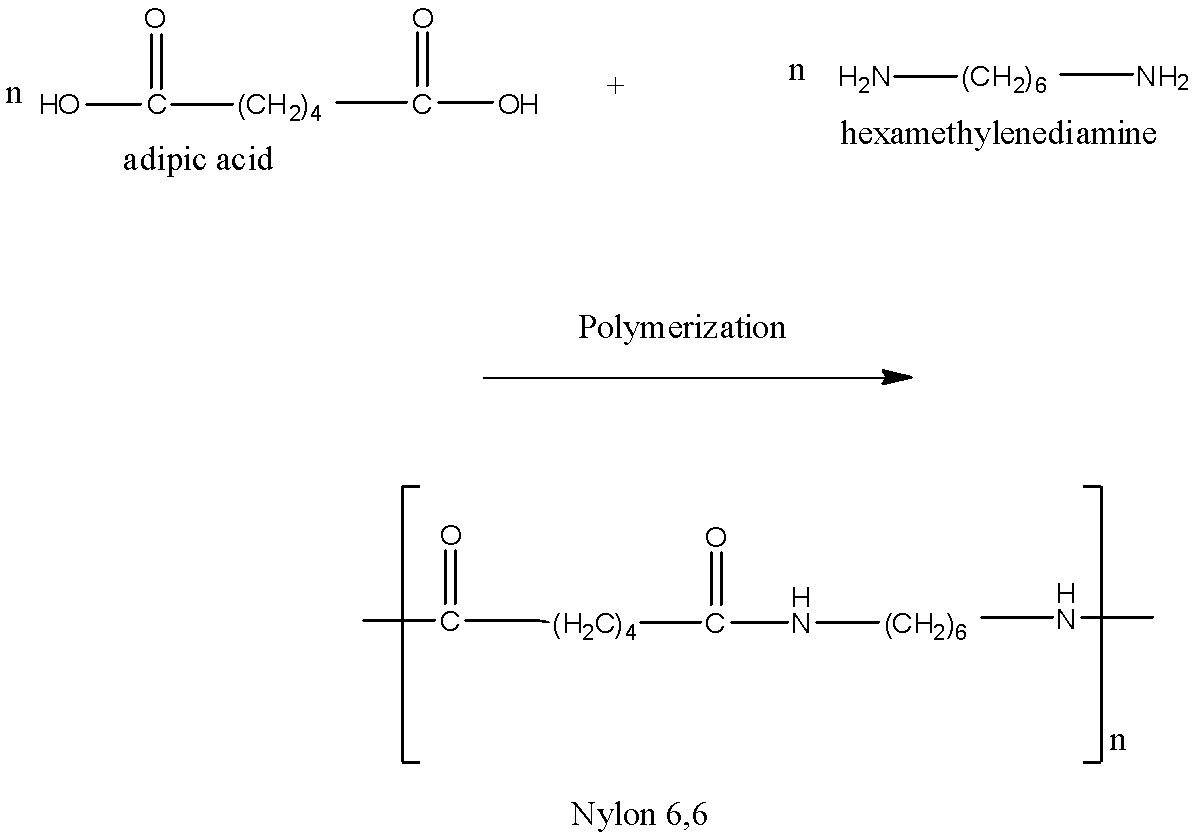
The two monomers for the synthesis of Nylon 6, 6 are:(a)- $HOOC{{(C{{H}_{2}})}_{6}}COOH,\\text{ }{{H}_{2}}N{{(C{{H}_{2}})}_{6}}N{{H}_{2}}$ (b)- $HOOC{{(C{{H}_{2}})}_{6}}COOH,\\text{ }{{H}_{2}}N{{(C{{H}_{2}})}_{4}}N{{H}_{2}}$ (c)- $HOOC{{(C{{H}_{2}})}_ ...

Chemical Recycling of End‐of‐Life Polyamide 6 via Ring Closing Depolymerization - Alberti - 2019 - ChemistrySelect - Wiley Online Library

Write the complete mechanism for the formation of Nylon-4,10 using sebacoyl chloride as one of the starting materials. You only have to show the formation of one amide bond. | Homework.Study.com

Use curved arrow notation to draw the mechanism for the formation of nylon-6 ,10 from sebacoyl chloride, 1,6-hexanediamineand sodium hydroxide. | Homework.Study.com